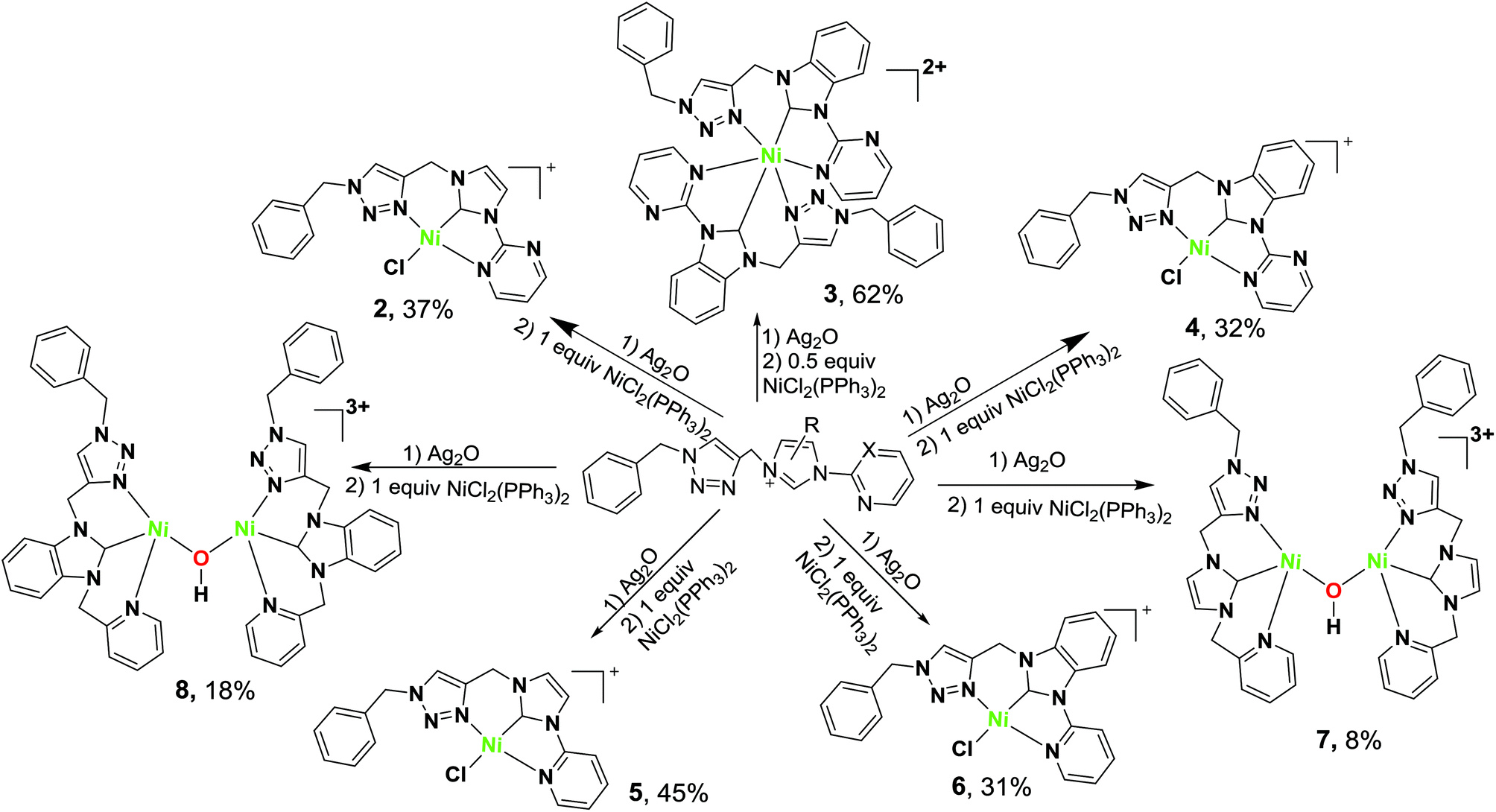
Unsymmetrical NCN-pincer mononuclear and dinuclear nickel( ii ) complexes of N-heterocyclic carbene (NHC): synthesis, structure and catalysis for Suzu ... - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03944H

Nickel(II)- and Palladium(II)-NHC Complexes from Hydroxypyridine Functionalized C,O Chelate Type Ligands: Synthesis, Structure, and Catalytic Activity toward Kumada–Tamao–Corriu Reaction | Organometallics

Nickel(II) Pincer Carbene Complexes: Oxidative Addition of an Aryl C–H Bond to Form a Ni(II) Hydride | Organometallics
![Ni(NHC)2] as a Scaffold for Structurally Characterized trans [H−Ni−PR2] and trans [R2P−Ni−PR2] Complexes - Sabater - 2021 - Chemistry – A European Journal - Wiley Online Library Ni(NHC)2] as a Scaffold for Structurally Characterized trans [H−Ni−PR2] and trans [R2P−Ni−PR2] Complexes - Sabater - 2021 - Chemistry – A European Journal - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/f9899e7f-8771-4ad0-b47c-06e193f2a547/chem202101484-toc-0001-m.jpg)
Ni(NHC)2] as a Scaffold for Structurally Characterized trans [H−Ni−PR2] and trans [R2P−Ni−PR2] Complexes - Sabater - 2021 - Chemistry – A European Journal - Wiley Online Library

Synthesis and structural characterization of nickel(II) complexes of 20-membered macrocyclic rings bearing chelating bis(N-heterocyclic carbene) ligands - ScienceDirect

Two-Coordinate d9 Complexes. Synthesis and Oxidation of NHC Nickel(I) Amides | Journal of the American Chemical Society

An Unsaturated Nickel(0) NHC Catalyst: Facile Preparation and Structure of Ni(0)(NHC)2, Featuring a Reduction Process from Ni(II)(NHC)(acac)2 | Organometallics
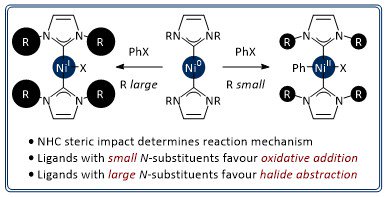
Steric effects determine the mechanisms of reactions between bis(N-heterocyclic carbene)-nickel(0) complexes and aryl halides
![Catalysts | Free Full-Text | Pyridine-Chelated Imidazo[1,5-a]Pyridine N-Heterocyclic Carbene Nickel(II) Complexes for Acrylate Synthesis from Ethylene and CO2 Catalysts | Free Full-Text | Pyridine-Chelated Imidazo[1,5-a]Pyridine N-Heterocyclic Carbene Nickel(II) Complexes for Acrylate Synthesis from Ethylene and CO2](https://www.mdpi.com/catalysts/catalysts-10-00758/article_deploy/html/images/catalysts-10-00758-g001.png)
Catalysts | Free Full-Text | Pyridine-Chelated Imidazo[1,5-a]Pyridine N-Heterocyclic Carbene Nickel(II) Complexes for Acrylate Synthesis from Ethylene and CO2

Nickel(II) N‐Heterocyclic Carbene Complexes: Versatile Catalysts for C–C, C–S and C–N Coupling Reactions - Junquera - 2017 - European Journal of Inorganic Chemistry - Wiley Online Library
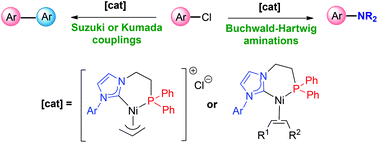
Phosphine-functionalized NHC Ni(ii) and Ni(0) complexes: synthesis, characterization and catalytic properties - Dalton Transactions (RSC Publishing)

Pincer‐Type Mesoionic Carbene Nickel(II) Complexes: Synthesis, Properties, Reactions, and Catalytic Application to the Suzuki–Miyaura Coupling Reaction of Aryl Bromides - Matsubara - 2022 - European Journal of Inorganic Chemistry - Wiley Online Library
![Catalysts | Free Full-Text | Pyridine-Chelated Imidazo[1,5-a]Pyridine N-Heterocyclic Carbene Nickel(II) Complexes for Acrylate Synthesis from Ethylene and CO2 Catalysts | Free Full-Text | Pyridine-Chelated Imidazo[1,5-a]Pyridine N-Heterocyclic Carbene Nickel(II) Complexes for Acrylate Synthesis from Ethylene and CO2](https://www.mdpi.com/catalysts/catalysts-10-00758/article_deploy/html/images/catalysts-10-00758-g001-550.jpg)
Catalysts | Free Full-Text | Pyridine-Chelated Imidazo[1,5-a]Pyridine N-Heterocyclic Carbene Nickel(II) Complexes for Acrylate Synthesis from Ethylene and CO2
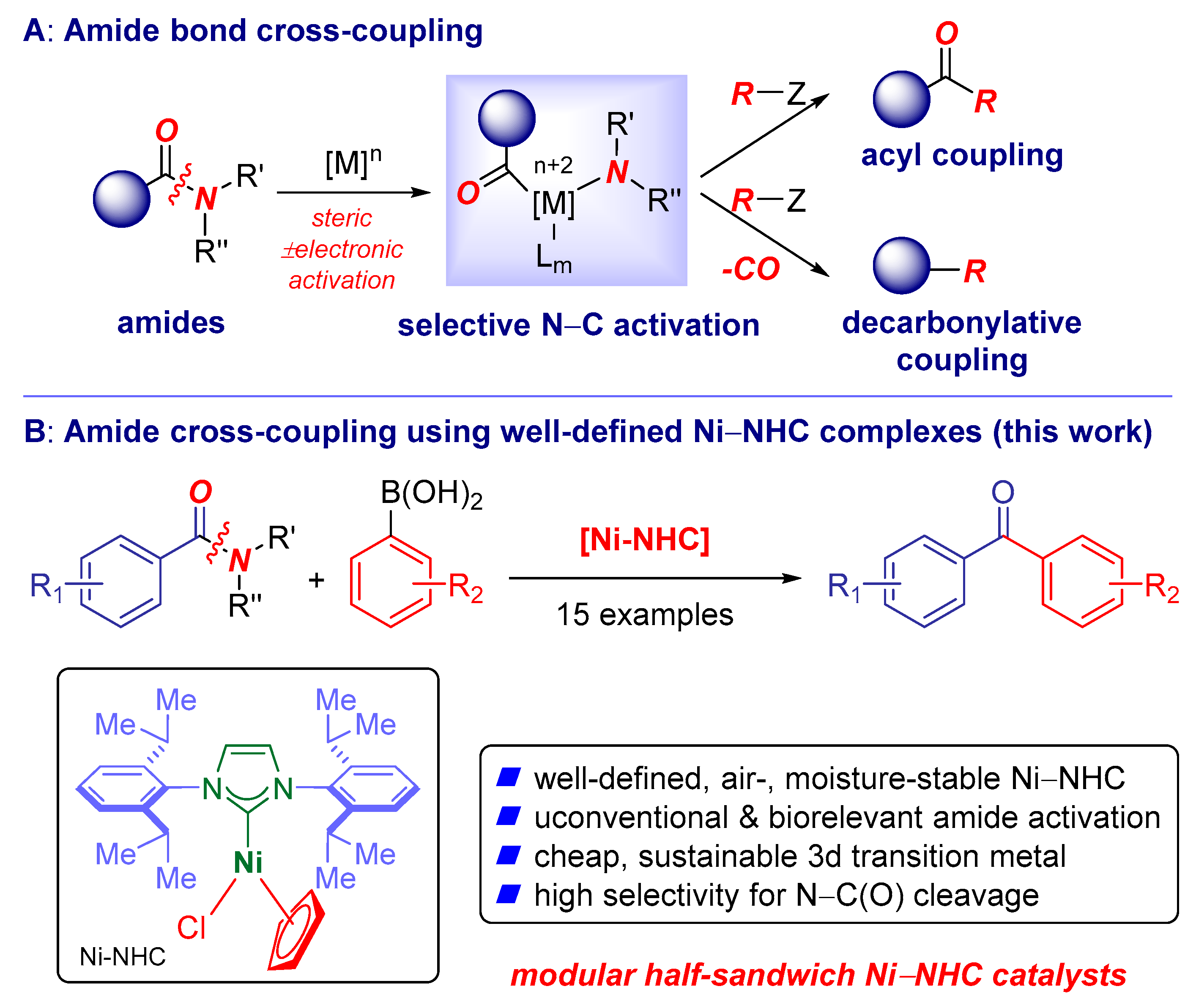
Catalysts | Free Full-Text | Suzuki–Miyaura Cross-Coupling of Amides Using Well-Defined, Air- and Moisture-Stable Nickel/NHC (NHC = N-Heterocyclic Carbene) Complexes
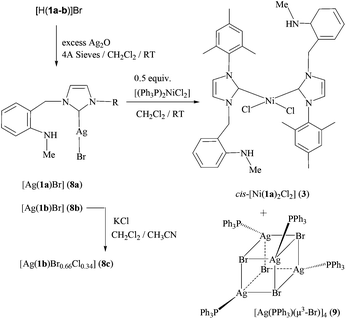
Direct synthesis of cis -dihalido-bis(NHC) complex of nickel( ii ) and catalytic application in olefin addition polymerization : Effect of halogen co- ... - Dalton Transactions (RSC Publishing) DOI:10.1039/C3DT50957E

Inorganics | Free Full-Text | Nickel(II) N-Heterocyclic Carbene Complex for the Hydrogenation of 2-Acetylpyridine under Mild Conditions

Unsymmetrical NCN-pincer mononuclear and dinuclear nickel(ii) complexes of N-heterocyclic carbene (NHC): synthesis, structure and catalysis for Suzuki–Miyaura cross-coupling - Dalton Transactions (RSC Publishing)
![Synthesis and Characterization of [(NHC)Ni(styrene)2] Complexes: Isolation of Monocarbene Nickel Complexes and Benchmarking of %VBur in (NHC)Ni-π Systems | Organometallics Synthesis and Characterization of [(NHC)Ni(styrene)2] Complexes: Isolation of Monocarbene Nickel Complexes and Benchmarking of %VBur in (NHC)Ni-π Systems | Organometallics](https://pubs.acs.org/cms/10.1021/acs.organomet.8b00394/asset/images/large/om-2018-00394b_0009.jpeg)
Synthesis and Characterization of [(NHC)Ni(styrene)2] Complexes: Isolation of Monocarbene Nickel Complexes and Benchmarking of %VBur in (NHC)Ni-π Systems | Organometallics

An Unsaturated Nickel(0) NHC Catalyst: Facile Preparation and Structure of Ni(0)(NHC)2, Featuring a Reduction Process from Ni(II)(NHC)(acac)2 | Organometallics
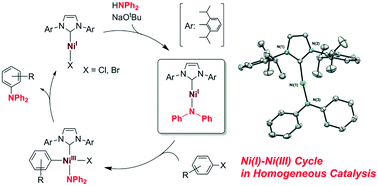


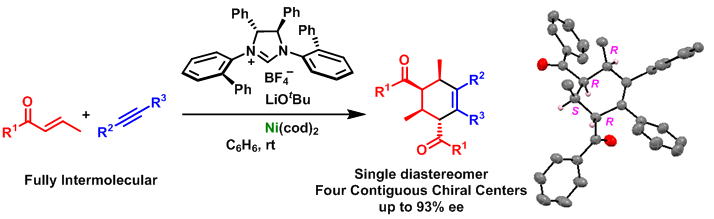
![NHC-Ni(II)-catalyzed cyclopropene-isocyanide [5 + 1] benzannulation | Nature Communications NHC-Ni(II)-catalyzed cyclopropene-isocyanide [5 + 1] benzannulation | Nature Communications](https://media.springernature.com/m685/springer-static/image/art%3A10.1038%2Fs41467-022-31896-y/MediaObjects/41467_2022_31896_Fig1_HTML.png)
